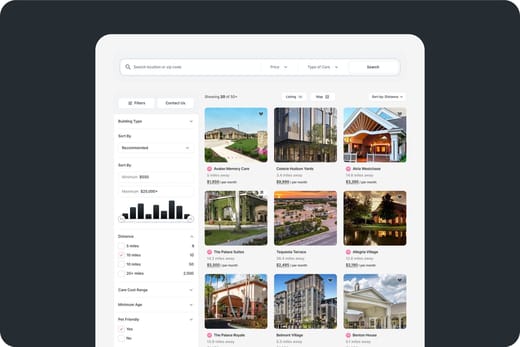
Deficiencies (last 6 years)
Deficiencies (over 6 years)
8.5 deficiencies/year
Deficiencies are regulatory violations found during state inspections.
63% worse than New Jersey average
New Jersey average: 5.2 deficiencies/yearDeficiencies per year
24
18
12
6
0
Occupancy
Latest occupancy rate
100% occupied
Based on a January 2026 inspection.
This facility has shown a steady increase in demand based on occupancy rates.
Occupancy rate over time
Inspection Report
Complaint Investigation
Census: 2
Capacity: 2
Deficiencies: 1
Date: Jan 2, 2026
Visit Reason
The inspection was conducted based on complaints NJ1875890 and 2681814 to investigate the facility's failure to develop and follow adequate fall prevention interventions for a resident at risk for falls.
Complaint Details
The complaint investigation found that the facility failed to develop and follow adequate fall prevention interventions for Resident #2, who was severely cognitively impaired and required substantial assistance. The resident fell when left unattended on a shower chair while staff went to get a towel. Interviews with staff and management confirmed that leaving the resident unattended was against facility policy and care plan requirements.
Findings
The facility failed to implement adequate fall prevention measures for Resident #2, who was at high risk for falls and required substantial assistance. The resident fell while left unattended on a shower chair, and staff did not follow care plan interventions or facility policies regarding supervision during bathing.
Deficiencies (1)
Failure to ensure a nursing home area is free from accident hazards and provide adequate supervision to prevent accidents.
Report Facts
Residents affected: 1
Census: 2
Total capacity: 2
Employees mentioned
| Name | Title | Context |
|---|---|---|
| CNA #1 | Certified Nursing Assistant | Named in fall incident where resident was left unattended on shower chair |
| CNA #2 | Certified Nursing Assistant | Interviewed regarding showering procedures and supervision |
| Unit Manager | Unit Manager | Interviewed regarding staff supervision policies during bathing |
| Director of Nursing | Director of Nursing | Interviewed regarding care plan expectations and staff actions |
| Licensed Practical Nurse #1 | Licensed Practical Nurse | Wrote progress note documenting fall incident |
Notice
Deficiencies: 0
Date: Nov 19, 2025
Visit Reason
This document serves to inform individuals about the privacy practices of NJDHSS, including how their medical information may be used and disclosed, and their rights related to this information.
Findings
The notice explains the types of information covered, reasons for use and disclosure of health information, individuals' rights to access and control their information, and the legal duties of NJDHSS to protect privacy.
Report Facts
Effective date: 2011
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Devon L. Graf | Director, Office of Legal and Regulatory Compliance | Listed as NJDHSS Privacy Officer contact for questions about the notice |
Inspection Report
Annual Inspection
Deficiencies: 9
Date: Apr 17, 2025
Visit Reason
The inspection was conducted as an annual survey to assess compliance with regulatory requirements including resident care, medication administration, infection control, and facility licensing.
Findings
The facility was found deficient in multiple areas including failure to obtain written consent for video monitoring, inadequate fall prevention interventions, improper catheter care, failure to post nurse staffing daily, medication administration errors, improper medication storage, failure to follow therapeutic diet orders, noncompliance with Medicaid resident admission requirements, and lapses in infection control practices during a COVID-19 outbreak.
Deficiencies (9)
Failure to ensure personal privacy and obtain written consent for video monitoring devices in resident rooms.
Failure to identify causal factors for falls and implement adequate interventions to prevent further falls for a cognitively impaired resident.
Failure to ensure Foley catheter drainage bag was stored off the floor to prevent urinary tract infections.
Failure to post daily nurse staffing information consistently.
Medication administration error: resident received incorrect dose of Hydralazine (25 mg instead of 50 mg).
Failure to properly label, store, and dispose of medications in medication carts including undated opened vials and improper refrigeration.
Failure to ensure residents received therapeutic diets as prescribed and special dietary instructions such as no straws were followed, resulting in immediate jeopardy for aspiration risk.
Failure to admit Medicaid-eligible residents per facility designation and regulatory requirements; facility admitted only short-term rehab residents and did not accept Medicaid residents.
Failure to implement infection prevention and control program adequately during COVID-19 outbreak including improper use of PPE, inadequate hand hygiene, and failure to disinfect shared medical equipment.
Report Facts
Residents affected: 2
Residents affected: 1
Residents affected: 1
Residents affected: 2
Residents affected: 2
Medications prepared: 10
Medication carts inspected: 8
Resident census: 121
Employees mentioned
| Name | Title | Context |
|---|---|---|
| LPN #4 | Licensed Practical Nurse | Named in medication administration and infection control deficiencies for improper hand hygiene and equipment disinfection |
| RN #1 | Registered Nurse/Unit Manager | Named in infection control deficiency for inconsistent PPE use and guidance |
| CNA #1 | Certified Nurse Aide | Named in infection control and therapeutic diet deficiencies for improper PPE use and providing thin liquids instead of nectar thickened liquids |
| Director of Nursing | Director of Nursing | Interviewed regarding video monitoring consent, fall prevention, medication errors, infection control |
| Licensed Nursing Home Administrator | Administrator | Interviewed regarding Medicaid admission policy and facility operation |
| Speech Therapist #1 | Speech Therapist | Interviewed regarding therapeutic diet and aspiration risk |
| Director of Dietary | Director of Dietary | Interviewed regarding therapeutic diet preparation and thickened liquids |
| Assistant Director of Nursing | Assistant Director of Nursing | Interviewed regarding fall prevention and medication administration |
| Registered Nurse/Unit Manager | Registered Nurse/Unit Manager | Interviewed regarding medication administration and infection control |
| Infection Preventionist Registered Nurse | Infection Preventionist | Interviewed regarding infection control practices and PPE use |
| Director of Social Services | Director of Social Services | Interviewed regarding resident discharge and Medicaid admission policies |
| Social Worker | Social Worker | Interviewed regarding Medicaid admission policies and resident discharge |
| LPN #1 | Licensed Practical Nurse | Named in infection control deficiencies for failure to disinfect equipment and improper PPE use |
| LPN #2 | Licensed Practical Nurse | Named in infection control deficiencies for improper PPE use |
Inspection Report
Routine
Census: 122
Capacity: 124
Deficiencies: 12
Date: Apr 17, 2025
Visit Reason
A Recertification/LSC survey was conducted from 4/10/25 through 4/17/25 to determine compliance with 43 CFR Part 483 requirements for Long Term Care Facilities, including complaint investigations.
Complaint Details
Complaint investigation was conducted based on multiple complaint numbers NJ 170566, NJ 170732, NJ 175978, NJ 177289, NJ 178091, NJ 178739, NJ 182695, NJ 183695. The complaint findings were substantiated as evidenced by multiple deficiencies cited.
Findings
The facility was found not in compliance with multiple regulatory requirements including dietary orders, personal privacy, ADL care, infection control, medication administration, emergency preparedness, and life safety code. Immediate Jeopardy was identified related to failure to ensure dietary orders and special instructions for residents at risk of choking and aspiration. Corrective actions and re-education plans were implemented and verified.
Deficiencies (12)
Failure to ensure dietary orders and special instructions for residents at risk of choking and aspiration, resulting in Immediate Jeopardy.
Failure to ensure personal privacy was maintained when implementing video monitoring devices inside resident rooms.
Failure to provide appropriate and timely personal hygiene care for dependent residents.
Failure to provide adequate supervision and assistance to prevent accidents and timely response to call lights.
Failure to ensure medication administration was conducted according to physician orders and nursing standards.
Failure to maintain emergency preparedness plan and documentation as required.
Failure to post nurse staffing data as required by regulation.
Failure to ensure infection prevention and control measures including proper use of PPE and hand hygiene.
Failure to ensure proper storage, labeling, and disposal of medications and biologicals.
Failure to ensure therapeutic diets were prescribed and followed according to physician orders.
Failure to comply with licensure requirements including Medicaid acceptance and admission agreements.
Failure to comply with life safety code requirements including fire door inspections and fire extinguisher maintenance.
Report Facts
Census: 122
Total Capacity: 124
Sample Size: 30
Deficiency Count: 12
Date Survey Completed: Apr 17, 2025
Plan of Correction Completion Date: May 13, 2025
Inspection Report
Complaint Investigation
Deficiencies: 1
Date: Apr 10, 2025
Visit Reason
The inspection was conducted based on a complaint alleging inadequate incontinence care and personal hygiene for residents at the facility.
Complaint Details
Complaint # NJ 170732. The complaint was substantiated based on observations, interviews, and record reviews confirming inadequate care and delayed staff response.
Findings
The facility failed to provide appropriate incontinence care and personal hygiene for multiple residents, including failure to timely assist residents with toileting, inadequate nail care, and delayed response to call lights. Observations and interviews confirmed residents were found with soiled briefs, long and jagged nails with debris, and delayed assistance leading to resident distress.
Deficiencies (1)
Failure to provide appropriate incontinence care and personal hygiene for 7 of 13 residents reviewed.
Report Facts
Residents affected: 7
Residents reviewed: 13
Resident units affected: 2
BIMS scores: 14
BIMS scores: 11
BIMS scores: 12
BIMS scores: 12
BIMS scores: 10
BIMS scores: 8
Employees mentioned
| Name | Title | Context |
|---|---|---|
| LPN #1 | Licensed Practical Nurse | Assisted Resident #15 with toileting and acknowledged nail care issues |
| Unit Manager | Unit Manager | Observed Resident #73's nails and discussed nail care responsibilities |
| CNA #1 | Certified Nurse Aide | Assisted Resident #322 with shower and acknowledged nail care deficiencies |
| LPN #2 | Licensed Practical Nurse | Discussed nail care importance and observed Resident #322's nails |
| RN/UM | Registered Nurse/Unit Manager | Discussed facility policy on nail care and acknowledged concerns |
| RN | Registered Nurse | Observed Resident #81's incontinence brief and discussed incontinence rounds |
| Director of Nursing | Director of Nursing | Met with survey team to discuss concerns |
| Licensed Nursing Home Administrator | Licensed Nursing Home Administrator | Met with survey team to discuss concerns |
Inspection Report
Complaint Investigation
Census: 122
Deficiencies: 0
Date: Jun 28, 2024
Visit Reason
The inspection was conducted as a complaint investigation based on multiple complaint numbers NJ00171568, NJ00171569, NJ00173253, and NJ00174253.
Complaint Details
The complaint investigation was based on four complaint numbers. The facility was found compliant with no deficiencies.
Findings
The facility was found to be in compliance with the requirements of 42 CFR, Part 483, Subpart B, for long term care facilities based on this complaint visit. No deficiencies were cited.
Report Facts
Sample size: 8
Inspection Report
Routine
Deficiencies: 5
Date: Dec 8, 2023
Visit Reason
The inspection was conducted to evaluate compliance with professional standards of quality, pharmaceutical services, medication administration, facility licensing, infection prevention and control, and other regulatory requirements at Total Rehab Moorestown.
Findings
The facility was found deficient in multiple areas including failure to clarify a physician's order for medication application, failure to promptly document controlled drug administration, failure to secure medication carts and properly label medications, failure to notify CMS and obtain authorization for a facility name change, and failure to follow proper infection control practices during medication administration.
Deficiencies (5)
Failed to clarify a physician's order for lidocaine patch application site from 10/25/23 until 11/30/23 for 1 of 4 residents observed during medication observation.
Failed to promptly record removal of controlled drugs from narcotic inventory for 1 of 3 nurses observed during medication pass.
Failed to secure medication administration cart during medication pass and maintain appropriate labeling/dating for medications on 2 of 8 medication carts inspected.
Failed to notify CMS and receive authorization for a change in the facility's name, using an unapproved name in signage and documents.
Failed to follow appropriate infection control practices by placing loose medication in the same cup with unopened packets and handling medications with ungloved hands.
Report Facts
Residents affected: 4
Residents affected: 3
Medication carts inspected: 8
Residents affected: 419
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Licensed Practical Nurse (LPN) | Administered medications to Resident #94 and #80; involved in medication documentation and administration deficiencies | |
| Director of Nursing (DON) | Interviewed regarding medication order clarification and narcotic documentation; provided in-service education | |
| Pharmacy Consultant (PC) | Provided information on medication labeling and expiration | |
| Licensed Nursing Home Administrator (LNHA) | Discussed facility name change and CMS notification issues | |
| Registered Nurse Infection Preventionist (RN/IP) | Interviewed regarding infection control practices | |
| Department Nurse Manager (DNM) | Observed handing medications with ungloved hands contributing to infection control deficiency |
Inspection Report
Deficiencies: 1
Date: Dec 8, 2023
Visit Reason
The inspection was conducted to evaluate the facility's compliance with professional standards of quality in medication administration, specifically regarding clarification of a physician's order for a lidocaine patch for pain management.
Findings
The facility failed to clarify a physician's order for a lidocaine patch from 10/25/23 until 11/30/23 for one resident, resulting in nurses applying the patch without documented specification of which knee to apply it to. The Treatment Administration Record did not require documentation of patch location, and the Director of Nursing acknowledged the order was incomplete and should have been clarified.
Deficiencies (1)
Failure to clarify a physician's order for lidocaine patch application location, resulting in undocumented administration site.
Report Facts
Residents Affected: 1
Date of physician's order: Oct 25, 2023
Date order clarification ended: Nov 30, 2023
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Licensed Practical Nurse (LPN) | Observed administering medication and interviewed regarding patch application | |
| Director of Nursing (DON) | Interviewed and stated the order should have been clarified and was incomplete |
Inspection Report
Annual Inspection
Census: 118
Capacity: 124
Deficiencies: 11
Date: Dec 8, 2023
Visit Reason
A Recertification Survey was conducted to determine compliance with 42 CFR Part 483, Requirements for Long Term Care Facilities.
Findings
Deficiencies were cited related to medication order clarification, pharmacy services, medication labeling and storage, facility name compliance, infection control, and life safety code violations including egress door locking, hazardous area enclosure, corridor door maintenance, smoke barrier door gaps, electrical receptacle safety, and generator maintenance.
Deficiencies (11)
Failed to clarify a physician's order for medication application location for Resident #94.
Failed to promptly record removal of controlled drugs from narcotic inventory for 1 of 3 nurses observed.
Failed to secure medication administration cart during medication pass and maintain proper labeling/dating of medications on carts.
Facility name displayed and used did not correspond with CMS licensed and approved name.
Failed to follow infection control practices by placing loose medication into the same medication cup with unopened packets.
Egress doors had locking devices that could restrict emergency use of exit doors.
Failed to ensure fire-rated doors to hazardous areas were self-closing and properly labeled.
Failed to maintain 49 of 120 corridor doors to resident rooms to close and provide smoke protection.
Failed to maintain smoke barrier doors to resist transfer of smoke due to gaps when closed.
Failed to ensure electrical outlet near water source was equipped with Ground-Fault Circuit Interrupter (GFCI).
Failed to provide remote manual stop station for generator and failed to document monthly load test transfer times.
Report Facts
Sample size: 27
Number of residents observed with medication order issue: 1
Number of nurses observed with narcotic record issue: 1
Number of medication carts inspected: 8
Number of doors with latching issues: 49
Facility licensed bed capacity: 124
Facility census: 118
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Licensed Practical Nurse (LPN) | Involved in medication order clarification and narcotic record deficiencies | |
| Director of Nursing (DON) | Interviewed regarding medication order clarification and narcotic record deficiencies | |
| Housekeeping Director (HD) | Interviewed and observed during life safety code deficiencies | |
| Licensed Nursing Home Administrator (LNHA) | Interviewed regarding facility name change and compliance | |
| Regional LNHA | Interviewed regarding CMS 855A application for name change | |
| Pharmacy Consultant (PC) | Interviewed regarding medication labeling and dating | |
| Registered Nurse Infection Preventionist (RN/IP) | Interviewed regarding infection control practices |
Inspection Report
Complaint Investigation
Deficiencies: 2
Date: May 24, 2023
Visit Reason
The inspection was conducted based on complaint #NJ164303 regarding medication administration and documentation practices at the facility.
Complaint Details
Complaint #NJ164303 involved failure to notify the physician of medication refusals and failure to document medication administration properly, including intravenous medications.
Findings
The facility failed to notify the physician when residents refused medications and did not follow its policies on documentation in medical records. Additionally, the facility failed to provide evidence that an intravenous medication was administered according to physician orders and did not follow medication administration policies.
Deficiencies (2)
Failure to notify the physician when a resident refused medications and failure to follow documentation policies for medication refusals for 2 residents.
Failure to provide evidence that intravenous medication was administered according to physician's order and failure to follow medication administration and intermittent infusion administration policies for 1 resident.
Report Facts
Residents affected: 2
Residents affected: 1
Medication refusal dates: 4
Medication refusal dates: 3
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Regional Director of Nursing | Regional Director of Nursing | Interviewed regarding physician notification policy and documentation. |
| Acting Clinical Director/Licensed Practice Nurse | Acting Clinical Director/Licensed Practice Nurse | Interviewed regarding medication refusal notification procedures. |
| Registered Nurse | Registered Nurse | Interviewed regarding physician notification and documentation of medication refusals. |
| Director of Nursing | Director of Nursing | Interviewed regarding intravenous medication administration and documentation. |
| Licensed Practical Nurse | Licensed Practical Nurse | Reported to DON about medication administration but failure to document in computer. |
Inspection Report
Complaint Investigation
Census: 90
Deficiencies: 3
Date: May 24, 2023
Visit Reason
The inspection was conducted in response to Complaint #NJ164303 to investigate allegations of noncompliance with staffing ratios, medication administration, and notification procedures at Promedica Total Rehab + (Moorestown).
Complaint Details
Complaint #NJ164303 involved allegations of inadequate staffing ratios, failure to notify physicians of medication refusals, and improper medication administration documentation. The complaint was substantiated with findings of deficiencies in these areas.
Findings
The facility was found deficient in maintaining required minimum direct care staff to resident ratios on multiple evening shifts. The facility failed to notify physicians when residents refused medications and did not follow its documentation policies. Additionally, there was a failure to document administration of medication for one resident, indicating possible missed medication doses.
Deficiencies (3)
Failed to maintain required minimum direct care staff to resident ratios on 7 of 14 evening shifts from 5/7/2023 to 5/20/2023.
Failed to notify physicians when residents refused medications for 2 residents and failed to follow documentation policies.
Failed to provide evidence that medication was administered according to physician's order and failed to follow medication administration and infusion policies for 1 resident.
Report Facts
Census: 90
Evening shifts deficient in CNA staffing: 7
Sample size: 4
Required CNAs on deficient shifts: 13
Actual CNAs on deficient shifts: 10
Inspection Report
Complaint Investigation
Census: 81
Deficiencies: 2
Date: Dec 3, 2021
Visit Reason
The inspection was conducted based on complaints NJ142054, NJ142662, NJ144197, and NJ149476 to investigate compliance with staffing ratios and resident assessments.
Complaint Details
Complaint investigation based on complaints NJ142054, NJ142662, NJ144197, and NJ149476. The facility was found deficient in staffing and resident assessment practices. The facility was ultimately found in compliance with 42 CFR, Part 483, Subpart B, for long term care facilities based on this complaint visit.
Findings
The facility failed to maintain required minimum staff-to-resident ratios for Certified Nurse Aides (CNAs) on multiple shifts, potentially affecting all residents. Additionally, the facility nursing staff failed to have a Registered Nurse complete a timely assessment for a resident after a fall, indicating deficient resident assessment practices.
Deficiencies (2)
Failure to ensure staffing ratios were met to maintain the required minimum staff-to-resident ratios as mandated by the state of New Jersey for 22 of 28 shifts reviewed.
Failure to have a Registered Nurse complete a timely assessment for a resident after a fall.
Report Facts
Census: 81
Shifts reviewed: 28
Shifts deficient: 22
Sample size: 5
Staffing ratios required: 11
Staffing ratios actual: 5
Inspection Report
Deficiencies: 1
Date: Aug 23, 2021
Visit Reason
The inspection was conducted to evaluate compliance with professional standards of clinical practice during medication administration at the nursing facility.
Findings
The facility failed to follow professional standards of clinical practice during medication administration for 1 of 4 residents observed, specifically administering a Lidocaine Patch 4% instead of the ordered 5%. The Licensed Practical Nurse did not identify the discrepancy prior to administration and acknowledged the need to notify the physician to clarify the order.
Deficiencies (1)
Failure to follow professional standards of clinical practice during medication administration, including administering Lidocaine Patch 4% instead of the ordered 5%.
Report Facts
Medications dispensed: 13
Residents observed: 4
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Licensed Practical Nurse (LPN) | Administered incorrect medication strength and acknowledged failure to see the 4% strength on the box | |
| Care Manager | Stated that nurses should perform three checks comparing medication to physician's order | |
| Director of Nursing (DON) | Stated that nurses should review physician's order and compare against medication prior to administration |
Inspection Report
Annual Inspection
Census: 81
Deficiencies: 2
Date: Aug 23, 2021
Visit Reason
A Recertification Survey was conducted to determine compliance with 42 CFR Part 483, Requirements for Long Term Care Facilities.
Findings
Deficiencies were cited related to failure to maintain required minimum direct care staff to resident ratios for the day shift and failure to follow professional standards of clinical practice during medication administration for one resident.
Deficiencies (2)
Facility failed to maintain the required minimum direct care staff to resident ratios for the day shift as mandated by the State of New Jersey.
Facility failed to follow professional standards of clinical practice during medication administration for 1 of 4 residents observed.
Report Facts
Census: 81
Deficiency count: 2
Staffing ratios not met: 13
CNA staffing counts: 8
Sign on bonus: 3000
Referral bonus: 500
Staffing bonus: 200
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Administrator | Stated there is a CNA shortage and described staffing efforts and bonuses | |
| Licensed Practical Nurse (LPN) | Observed administering medication incorrectly during medication pass | |
| Care Manager | Stated nurses should perform three checks during medication administration | |
| Director of Nursing (DON) | Stated nurses should review physician orders and perform checks prior to medication administration |
Inspection Report
Routine
Census: 81
Deficiencies: 0
Date: Aug 23, 2021
Visit Reason
A COVID-19 Focused Infection Control Survey was conducted by the New Jersey Department of Health to assess compliance with infection control regulations.
Findings
The facility was found to be in compliance with 42 CFR §483.80 infection control regulations and has implemented the CMS and CDC recommended practices for COVID-19.
Report Facts
Sample size: 5
Inspection Report
Life Safety
Deficiencies: 1
Date: Aug 20, 2021
Visit Reason
A Life Safety Code Survey was conducted by the New Jersey Department of Health, Health Facility Survey and Field Operations to assess compliance with Medicare/Medicaid participation requirements related to Life Safety from Fire and the 2012 Edition of the NFPA 101 Life Safety Code for existing health care occupancies.
Findings
The facility was found to be in noncompliance due to failure to provide battery backup emergency lighting in the main electrical room above the emergency generator's transfer switch, independent of the building's electrical system and emergency generator.
Deficiencies (1)
Facility failed to provide a battery backup emergency light in the main electrical room above the emergency generator's transfer switch, independent of the building's electrical system and emergency generator.
Report Facts
Smoke zones: 15
Building stories: 3
Employees mentioned
| Name | Title | Context |
|---|---|---|
| Maintenance Director | Verified the deficiency of missing emergency lighting in the main electrical room during inspection. |
Inspection Report
Routine
Census: 85
Deficiencies: 0
Date: Mar 9, 2021
Visit Reason
A COVID-19 Focused Infection Control Survey was conducted by the New Jersey Department of Health to assess compliance with infection control regulations.
Findings
The facility was found to be in compliance with 42 CFR §483.80 infection control regulations and has implemented the CMS and CDC recommended practices for COVID-19.
Report Facts
Sample size: 8
Sample size: 3
Inspection Report
Routine
Census: 75
Deficiencies: 0
Date: Jan 25, 2021
Visit Reason
A COVID-19 Focused Infection Control Survey was conducted by the New Jersey Department of Health to assess compliance with infection control regulations.
Findings
The facility was found to be in compliance with 42 CFR §483.80 infection control regulations and has implemented the CMS and CDC recommended practices for COVID-19.
Report Facts
Sample size: 3
Inspection Report
Routine
Census: 89
Deficiencies: 0
Date: Nov 20, 2020
Visit Reason
A COVID-19 Focused Infection Control Survey was conducted by the New Jersey Department of Health to assess compliance with infection control regulations and recommended practices for COVID-19.
Findings
The facility was found to be in compliance with 42 CFR §483.80 infection control regulations and has implemented the CMS and CDC recommended practices for COVID-19.
Report Facts
Sample size: 6
Viewing
Loading inspection reports...